詢 價
索取COA
產(chǎn)品描述
產(chǎn)品數(shù)據(jù)庫
| Introduction | |
| Format | Genomic DNA |
| Description | N/A |
| Technical Data | |
| DNA Change | c.9090delA |
| AA Change | p.T3033fs*29 |
| Mutation type | Deletion - Frameshift |
| Zygosity | Heterozygous |
| Allelic Frequency | 50.00% |
| Transcript | ENST00000380152 |
| Cosmic ID | N/A |
| Chr position(GRCh37) | chr13:32954023 |
| Buffer | Tris-EDTA |
| Product Information | |
| Intended Use | Research Use Only |
| Unit Size | 1ug |
| Concentration | Download for COA |
| Purofication | Download for COA |
| DNA electrophoresis | Download for COA |
| Sanger sequencing | 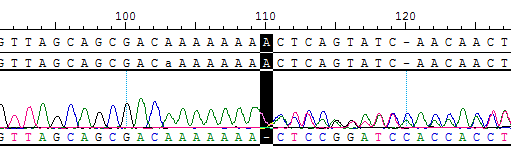 |
| Storage | 2-8°C |
| Expiry | 36 months from the date of manufacture |